Abstract
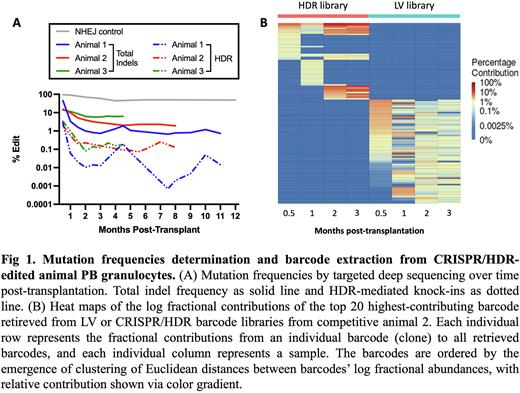
For precise genome editing via CRISPR/homology-directed repair (HDR), effective and safe editing of long-term engrafting hematopoietic stem cells (LT-HSCs) requires both sufficient HDR efficiency and protection of LT-HSC function and number. Loss or damage of LT-HSCs with resultant oligoclonal hematopoiesis can increase the risk of malignant transformation or long-term hematopoietic and immune dysfunction. HDR has adverse impacts on LT-HSC function as revealed by ex vivo studies and murine xenograft models, likely due to p53 activation, intracellular innate immune activation, and/or other pathways. However, the impact of HDR on true LT-HSC clonal dynamics in a relevant large animal model has not previously been studied. We tracked the clonal dynamics of CRISPR/HDR-edited barcoded HSPCs following autologous transplantation in rhesus macaques (RMs). HDR-favored gRNAs targeting the CD33 gene locus and an ssODN containing a 6 bp barcode ID and 14 bp random barcode sequences and flanked by CD33 homology arms were designed. Autologous CD34+ cells were electroporated with the Cas9 RNP and HDR cassette in the presence of i53 mRNA to inhibit p53-mediated cell death and reinfused into animal 1 following myeloablation. In animal 1, CD33 KO neutrophils were prevalent early following engraftment and then rapidly decreased over the next two months. Similar to CD33 expression level, we revealed that HDR knock-ins as well as total indel frequency drastically decreased following transplantation. This was in marked contrast to macaques transplanted with CD34+ HSPCs edited at the CD33 locus only for NHEJ, without inclusion of an HDR cassette (Kim et al, Cell, 2018, and Figure 1 below).
We next performed a competitive transplantation comparing clonal dynamics of edited cells to barcoded lentiviral (LV)-transduced cells in two macaques. For each animal, CD34+ cells were fractionated into 2 aliquots, and half were transduced with a barcoded GFP-expressing LV vector and half electroporated with the Cas9 RNP and HDR cassette with higher concentrations of i53 mRNA to enhance protection of CRISPR edited LT-HSCs. The two cell populations were combined and reinfused into myeloablated animals. CD33 KO cells in both animals decreased rapidly, however the higher concentration of i53 mRNA facilitated a less steep reduction in CD33 total indel frequency in vivo over time. However, HDR in these two animals also markedly decreased, similar to the pattern in animal 1. In contrast, LV-transduced GFP+ cell engraftment stabilized at 20% after 2 months. We retrieved embedded barcodes from CRISPR/HDR cells and revealed that a diversity of HDR clones contributed to early hematopoietic reconstitution, with restriction to a much smaller set of long-term clones at steady state, proportional in the 3 animals to the level of HDR overall. The diversity of HDR-edited contributing clones was far less than LV-transduced contributing clones long-term post-transplantation.
In conclusion, CRISPR/HDR edited cells were found to decrease rapidly after autologous transplantation in rhesus macaques despite substantial gene editing assessed in the infusion product prior to infusion, in contrast to much more stable LV-transduced cells. Clonality of HDR-edited cells drastically shrank at early stage and then relied on several dominant clones, which was somewhat improved by use of high dose i53 mRNA during editing. It will be important to find improved approaches to retain LT-HSC functional activity during the HDR editing process, or explore targeted gene correction approaches such as base or PRIME editing that may better preserve HSC activity.
Disclosures
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal